Clinical engineering roots
Early cardiac technology programs established a culture of clinician co-design, careful documentation, and measured device performance.

Reliable hemodynamic insight, remote care integration, and field service discipline for cardiac OR, ICU, step-down, and hospital-at-home programs.
Every Edwards Lifesciences program is framed for capital committees that need lifecycle cost, training readiness, and post-market response in one dependable plan.
Early cardiac technology programs established a culture of clinician co-design, careful documentation, and measured device performance.
Hospital teams began standardizing data review, interface validation, and device update windows across critical care departments.
Field support now includes FHIR observation mapping, SBOM review, service escalation rules, and training refreshers for dispersed care settings.

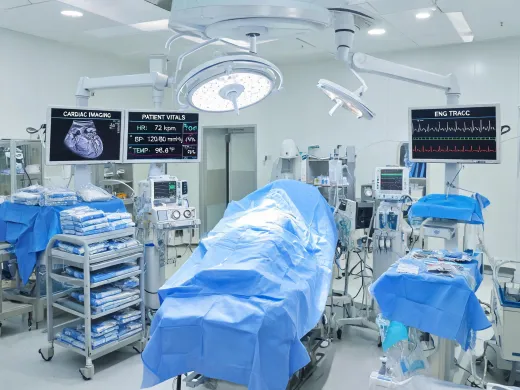


"The strongest monitoring programs are not just screens at the bedside; they are training, interface governance, service response, and procurement evidence working together."
Value Analysis Director, regional cardiac network
Ask for device documentation, service coverage, training resources, and remote-care integration notes in a single review packet.