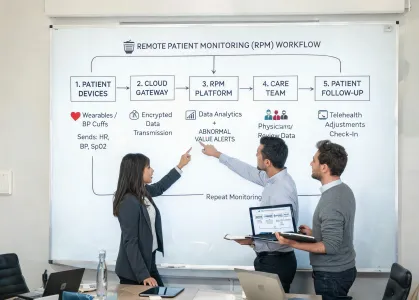
Mission
Make critical-care monitoring programs easier to trust at the bedside and easier to defend under FDA, CE MDR, and hospital value analysis review. Our teams focus on measurement consistency, service readiness, documentation discipline, and user training that holds up during a 3 a.m. escalation.